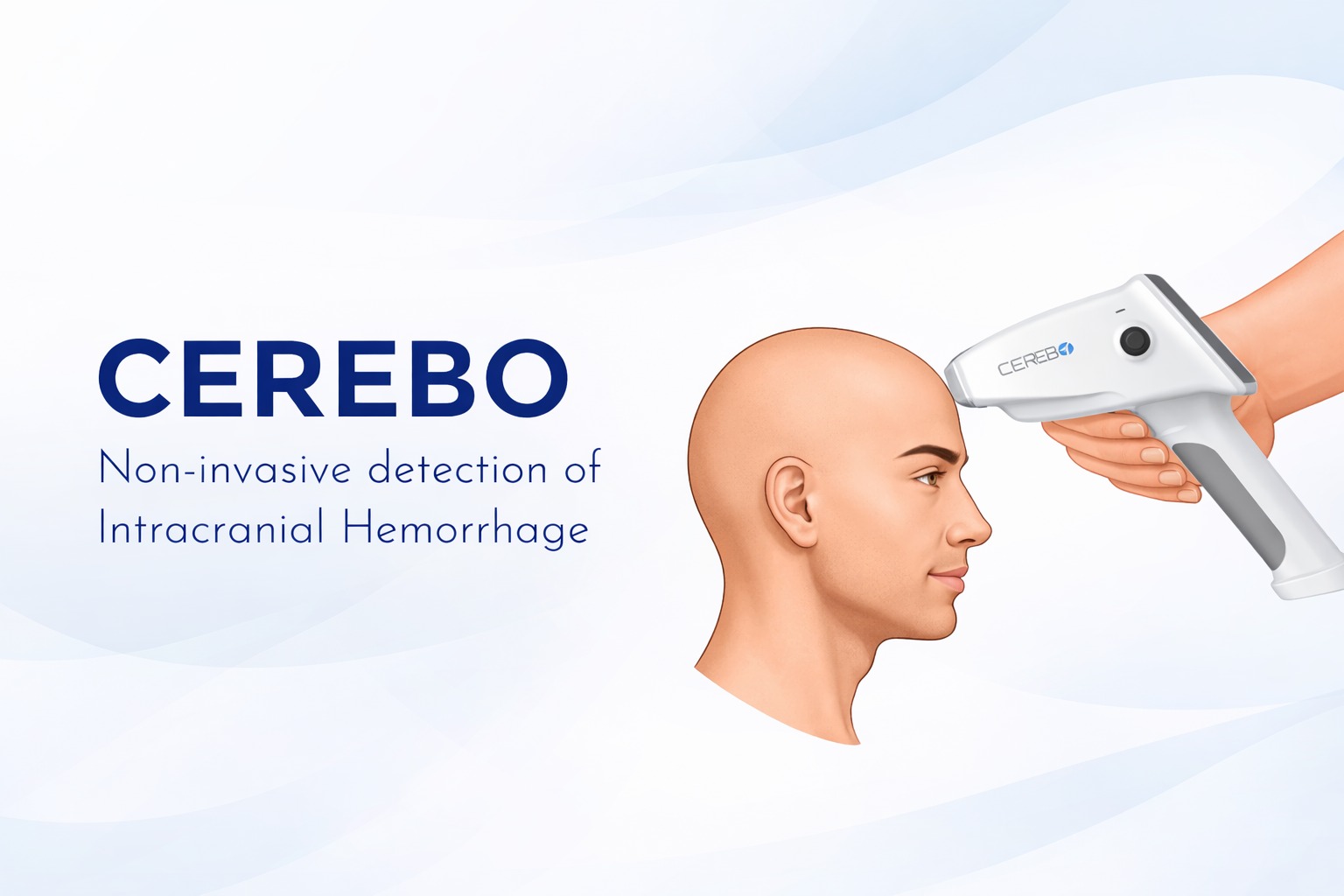
Cerebo: Portable Brain hemorrhage Detector
The Innovation Overview
CEREBO® is a non-invasive, machine-learning-powered diagnostic system designed to detect intracranial hemorrhages within the "Golden Hour." By engineering a handheld NIRS (Near-Infrared Spectroscopy) device, Techshlok helped Bioscan Research achieve a 97% clinical sensitivity in triaging Traumatic Brain Injury (TBI) patients—bypassing the need for immediate, high-cost CT/MRI scans in emergency settings.
The Client
Bioscan Research Pvt Ltd A deep-tech medical diagnostic leader, Bioscan Research is focused on non-invasive screening. Their goal was to democratize access to neuro-diagnostics, moving from expensive hospital infrastructure to affordable, portable point-of-care solutions.
Problem Statement
Bioscan’s core IP a method for detecting blood absorption signatures using NIR light was functionally proven in a laboratory setting but commercially unviable. To reach clinical deployment, three major "Real-World" barriers existed:
The Scalability Barrier: Laboratory-grade optical sensors were too bulky and sensitive for handheld use.
The Signal-to-Noise Ratio: In a portable device, the electromagnetic noise from the battery and processor threatened the integrity of sub-millivolt brain-signal data.
Diagnostic Reliability: The architecture had to work across all patient demographics (infants to elderly) without requiring clinical preparation (like head-shaving).
Solution
We re-engineered the laboratory setup into a Medical-Grade Integrated System. Our approach focused on "Hardening" the electronics for clinical environments:
- Miniaturized Optical Path: We architected a 3.5cm sensor-probe geometry that sitting between hair follicles, ensuring light reaches the required 3.0cm cranial depth for reliable hematoma detection.
ML-Edge Integration: The system was designed to process complex math-based algorithms locally on the device, providing an objective "Red/Green" alert in under 2 minutes.
Power Integrity Engineering: We developed ultra-stable voltage regulation to prevent diagnostic drift, ensuring the device remains accurate from the first patient to the last on a single charge.
Digital Reporting (The Cerebo Cloud): We integrated a seamless sync feature that generates instant PDF reports via Bluetooth/Cloud, allowing paramedics to transmit data to neurosurgeons while the patient is still in transit.
Overcoming the "Signal Noise"The primary technical victory was managing Internal Optical Interference. In a miniaturized device, the proximity of the processor to the NIRS sensors creates electromagnetic and thermal noise that can mimic a "bleed" signal.
- Hardware Hardening: Techshlok implemented advanced shielding and multi-layer PCB isolation to protect the sub-millivolt sensor signals.
- Zero-Radiation Design: Unlike CT scans, our architecture is 100% radiation-free, making it safe for repeated use on pregnant women and infants a massive regulatory win.
Business & Global Impact
The collaboration between Bioscan and Techshlok resulted in one of the most decorated medical devices in the Indian startup ecosystem:
- Clinical Validation: Multi-center studies at NIMHANS and AIIMS confirmed a 94.1% overall accuracy.
- Global Recognition: The product has secured 50+ awards worldwide, including accolades from Qualcomm, TATA Trusts, and NASSCOM
- Regulatory Milestone: Successfully developed into a TRL-6 computerized system, safe for use on high-risk groups like pregnant women and infants due to its 100% radiation-free design.
- Mass Production Ready: Transitioned from a fragile POC to a durable, manufacturable product now part of the ICMR (Indian Council of Medical Research) national registry.
A future-proof, intuitive design with excellent engineering.
